Type 2 diabetes mellitus (T2DM) is a polygenic metabolic disease characterized by elevated blood sugar levels due to pancreatic beta-cell functional impairment and insulin resistance in tissues such as skeletal muscle, adipose tissue and the liver (1). Millions of people around the globe are diagnosed with diabetes, and its incidence is estimated to double by 2030. It has become one of the most challenging public health issues of 21st century and the fifth leading cause of death worldwide (2). The American Diabetes Association (ADA) estimates that 1.7 million Americans are diagnosed with diabetes every year, which means about 4,660 new diagnoses of diabetes daily (3). Of the 30 million people who currently have diabetes in the United States, about 90% have T2DM.
This chronic disease process is accompanied by complications in various vital organs and can be associated with a higher risk for cardiovascular events, neurological changes, kidney impairment, osteoporosis, cognitive impairment (CI) and dementia (4). Though diabetes has a strong genetic component, as it tends to run in families, environment also has a significant role in triggering this condition. The notable risk factors for developing diabetes are obesity, advance in age, and sedentary lifestyle with lack of physical activity. However, many people with these risk factors do not develop diabetes and research studies indicate that complex interaction between genes and environment through epigenetic modifications makes a person susceptible to develop diabetes (5). Epigenetic mechanisms and their role in the development of diabetes and the significance of the environment in shaping the epigenome of an individual is presented in this article.
Epigenetic mechanisms
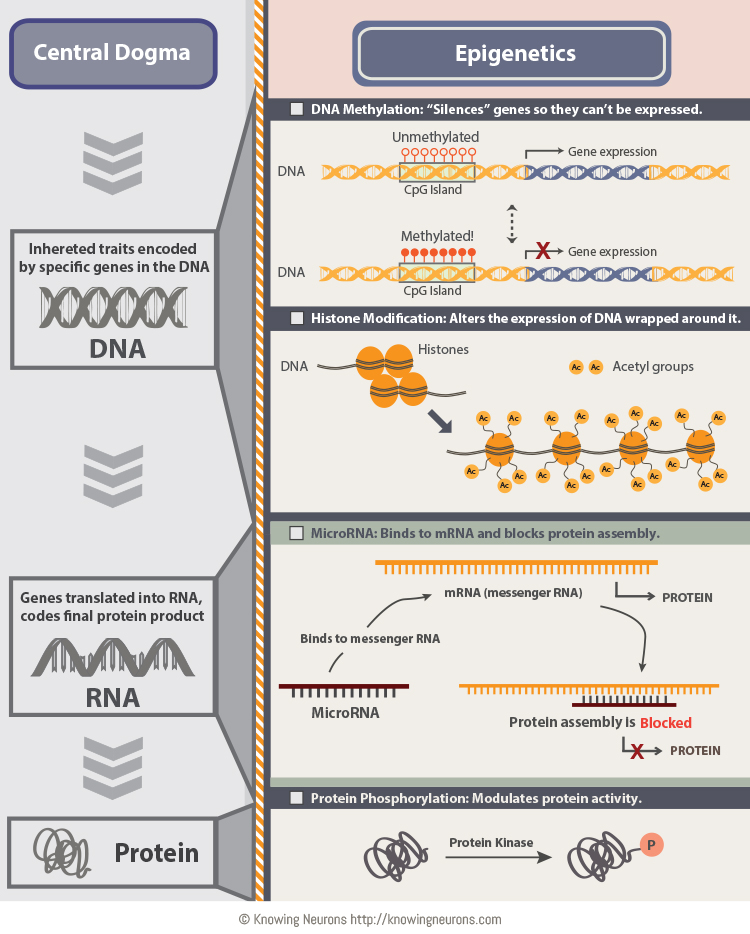
The notable epigenetic mechanisms that can alter gene expression are DNA methylation, histone modification, and non-coding RNA-mediated pathways (Figure 1). Briefly, in DNA methylation, a methyl group is added at the 5-carbon of the cytosine to form 5-methylcytosine. DNA methylation generally results in gene silencing or reduced gene expression (9). Another epigenetic mechanism which plays an important role in gene switching ‘on’ and ‘off’ is histone modification. Histones are globular proteins around which DNA coils to form nucleosomes. Histone modifications occur by enzyme catalyzed reactions such as lysine acetylation, lysine/arginine methylation, serine/threonine phosphorylation, and lysine ubiquitination/SUMOylation. These modifications in histones bring significant alterations in their functions (9) resulting in promotion or repression of gene transcription (10). The third epigenetic mechanism in regulating gene expression is micro RNAs. MicroRNAs (miRNA) are a class of non-coding single stranded RNAs of 19-25 nucleotides in length, which are reported to have a key role in the regulation of gene expression (5) (Figure 1).
Genes, Environment and Epigenetics
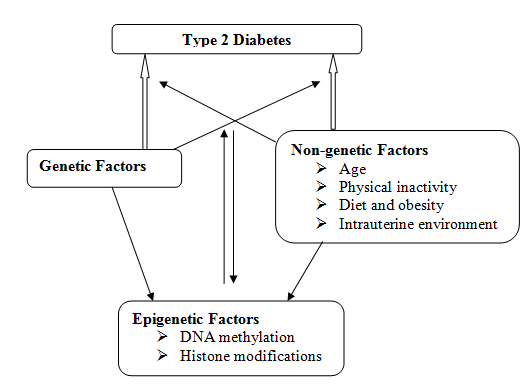
Though many genetic variants are shown to contribute to the development of T2DM, to date only PPARG, KCNJ11 and TCF7L2 are established genes associated with common forms of T2DM (12). Genome-wide association studies (GWAS) of recent times are helping in detecting and identifying more genes responsible for T2DM and could provide a complete list of genetic variants associated with this disease in the future (13). Diabetes is multifactorial disease indicating its emergence in individuals with genetic predisposition that depends on other factors.
Environment is reported to play a significant role in causing diabetes. Many environmental factors are known to cause changes in gene expression through epigenetic modifications (5)(14), such as altered DNA methylation or histone modifications. It is proposed that the environmental factors activate an intracellular signal, which, in turn marks the exact chromatin site for epigenetic modifications (14)(15) resulting in altered gene expression.
Some of the environmental factors occurring during embryogenesis (such as maternal diet and intrauterine nutrition) and early development could impact health and disease states even in adulthood (16). In addition to these, exposure to heavy metals, pesticides, cigarette smoking, and even deficiencies of some nutrients (folate and methionine) can stimulate alteration in epigenetic pathways (8). Additionally, obesity and age are also shown to alter these epigenetic mechanisms and might result in T2DM (5).
Epigenetics of Diabetes
In recent years many reports strongly point to the critical role of epigenetic modifications in the development and pathogenesis of cancer, asthma, arthritis, hypertension, etc. Lately epigenetic modifications are also implicated in the development of T2DM (Figure 2). Genes involved in the production and release of insulin are reported to be altered in diabetic islets compared to non-diabetic ones.
Analysis of pancreatic beta cells from diabetic and healthy individuals revealed epigenetic changes in approximately 850 genes with a fold change between 5-59% (1). Over 100 of the genes also had altered expression and many of these could contribute to reduced insulin production. About 17 candidate genes for type 2 diabetes including TCF7L2, THADA, KCNQ1, FTO, and IRSI were reported to be differentially methylated in pancreatic islets of type 2 diabetic population. Increased expression and decreased methylation of CDKN1A and PDE7B gene in type 2 diabetes was reported to result in impaired glucose-stimulated insulin release. This observation provides an evidence of diabetes associated epigenetic modifications and associated impaired insulin release (1).
Another gene UNC13B located on chromosome -9, is found to be hyper-methylated in diabetic patients and hyperglycemia upregulates its expression and is implicated in diabetic nephropathy (20).
MicroRNAs (miRNA) are also shown to be involved in glucose homeostasis and diabetes. One of the miRNAs specific to pancreatic islets is miR-375. Over-expression of this miRNA is found to reduce glucose-stimulated insulin release while its inhibition resulted in increase in insulin secretion (21). An inverse correlation between miRNA-192, miRNA-9 and insulin secretion was also reported, indicating that miRNAs may play a role in diabetes.
Histone modifications are also reported in diabetic patients. An in vitro study showed increased histone acetylation which in turn promoted inflammatory gene expression (22). Lastly, hyperglycemia induced histone modifications and DNA methylation of pro-inflammatory genes triggering the vascular inflammation are also reported (23). Thus emergence of diabetes in an individual is quite complex and results from the complex interaction between genes and environment. Many studies point to epigenetic modifications in mediating this interaction.
Conclusions
Diabetes is a complex metabolic disorder characterized by hyperglycemia, insufficient insulin secretion and insulin resistance. Incidence of diabetes is growing at an alarming rate at about 4660 new cases per day in the US. This disease runs in families, indicating that it has a genetic origin and genome wide association studies indicate about 100 gene variants associated with this disease. Though genes predispose a person to develop this disease, environment also contributes to its prevalence. The risk factors for diabetes include sedentary lifestyle, obesity and aging. Thus emergence of diabetes in an individual is quite complex and results from the complex interaction between genes and environment. Many studies point to epigenetic modifications (DNA methylation and histone modifications) in mediating this interaction.
References:
- Dayeh T, et al., (2014). Genome –wide DNA methylation analysis of human pancreatic islets from Type 2 Diabetic and Non-diabetic donors identifies candidate genes that influence insulin secretion. PLOS/Genetics, 10(3).
- Shaw JE, et al., (2010). Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract, 87: 4-14.
- American Diabetes Association. Fast facts: data and statistics about diabetes.
- Mastro A., et al., (2014). Cognitive impairment and dementia in Type 2 diabetes mellitus. US Pharm. 2014;39(10):33-37.
- Ling C., Groop L. (2009). Epigenetics: a molecular link between environmental factors and type 2 diabetes. Diabetes 56: 2718-2725.
- Bird A. (2007) Perceptions of epigenetics. Nature, 447:396-398.
- Accessed from http://learn.genetics.utah.edu/content/epigenetics/
- Epigenetics and Environmental Diseases. http://oehha.ca.gov/public_info/lecture/epigenetics.pdf
- Bernstein BE et al., (2007). The mammalian epigenome. Cell, 128: 669-681.
- Li B. et al., (2007). The role of chromatic during transcription. Cell, 128: 707-719.
- Knowing Neurons. (2013). Your Brain on Epigenetics [Web Graphic]. Retrieved from http://knowingneurons.com/2013/06/13/your-brain-on-epigenetics/
- Zeggini E., (2007). A new era for Type 2 diabetes genetics. Diabet Med, 24: 1181-1186 .
- Editorial (2007). Genomics of common diseases. Nature genetics, 39: 569.
- Bramswig, NC., et al., (2012). Epigenetics and diabetes treatment:an unrealized promise? Trends Endocrinol Metab.23(6): 286-291.
- Berger SL. et al., (2009) An operational definition of epigenetics. Genes Dev. 2009:23: 781-781.
- Roseboom T.J., (2001). Effects of prenatal exposure to the Dutch famine on adult disease in later life: an overview. Molecular and cellular endocrinology 185: 93-98.
- Toperoff G., et al., (2012). Genome wide survey reveals predisposing diabetes type 2-related DNA methylation variation in human peripheral blood. Hum Mol Genet, 21:371-383.
- Ling C., et al., (2008). Epigenetic regulation of PPARGC1A in human type 2 diabetic islets and effect on insulin secretion. Diabetologia, 51 : 615-622.
- O. Alibegivic AC., et al., (2010). Insulin resistance induced by physical inactivity is associated with multiple transcriptional changed in skeletal muscle in young men. American Journal of Phsyiology, Endocrinology and Metabolism, 299 (5): 572-563.
- Accessed from http://www.genecards.org/cgi-bin/carddisp.pl?gene=unc13B
- Poy, M.N., et al., (2004). A pancreatic islet-specific microRNA regulated insulin secretion. Nature 432(7014):226-230.
- Villeneuve L.M., et al., (2010). The role of epigenetics in the pathology of diabetic complications. American J Physiology: Renal Physiology, 299 (1): 14-25.

